The first is for Roger Green, who said he liked chemistry. He's also a trivia genius, so I tried to think of something he'd never heard of.
 |
| Chirality is all about "handedness." Bet you didn't know that a molecule could have a left or a right "hand"! (Image from Nature Publishing Group, Chiral molecules: High-speed photography, research highlight.) |
In my last ABC Wednesday post, I invited readers to share their favorite science topics. Here's a schedule so far:
Roger, chemistry, "C"
Leslie, oceanography, "D"
photowannabe, animals, "E"
PhenoMenon, astronomy, "F"
Carver, conservation of natural resources, "G"
ChrisJ, botony, "H"
LC Scotty, statistical mechanics, "I"
So, if there's a subject you're interested in, let me know.
Chirality is an important part of chemistry, particularly organic chemistry, the study of the chemistry of molecules with carbon in them.
With very simple molecules like H2O - water - there's only one way to arrange the three atoms - 2 hydrogens (H) and 1 oxygen (O) - in the molecule.
But with molecules with many atoms in them, you can have the same formula but not the same arrangement. Molecules can fit in a different order.
Chirality defines groups of molecules that are very, very similar, even in their arrangement. The only difference is that they are perfect mirror images of each other.
How can two things have a similar arrangement and be perfect mirror images, but not the same thing? Think of your hands. Your fingers and thumbs on each hand are in similar places, but your left and right hand are not the same. They're perfect mirror images of each other.
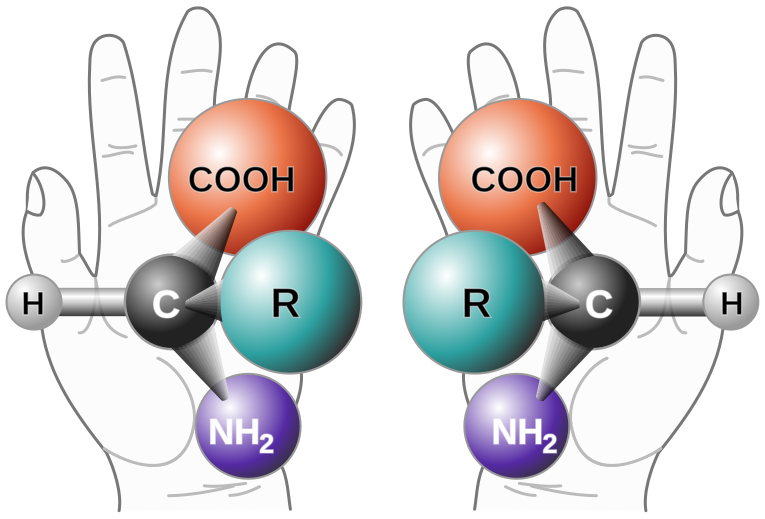 |
| A good image from Wikipedia showing the concept of chirality. |
 |
| A real molecule that shows chirality. Both of these are different orientations of alanine. The formula is the same for both, HOCCH(NH)CH. |
Next post is "D" on oceanography... trust me, you'll like it! :-) )
YOU HAVE SUCCEEDED! No, I didn't know the word, but the handedness description was useful. Hmm, I used that word handedness recently in a blog comment, but at 2:45 a.m., and can't sleep, can't say where.
ReplyDeleteNuts, I should have asked for cats...
ROG, ABC Wednesday
Of course, one can't build a nine-letter SCRABBLE word unless one builds it on an existing word, such as hi or chi or, as noted, chiral.
ReplyDeleteTrue! And like I said, sure to wow your friends. Not with points necessarily, but definitely with vocab.
DeleteYou're also a "trivia" genius, not a "trivial" genius. :-D Fixed it.
I DO appreciate the correction. (Although I MAY be a trivial genius too.)
ReplyDelete:-) Hey, don't knock being a trivial genius. I've made a career out of it! :-D
DeleteWow! I wish you'd been my Science teacher in school - I just might have gone further in it than I did. Great post!
ReplyDeleteLeslie
abcw team
That is seriously probably one of the nicest things anyone has ever said to me. :-) Thank you! And thanks for visiting!
DeleteOh I say - how intellectual - a librarian, a scientist and 2 teachers - Leslie and myself! Phew! We are quite a crowd eh? My favourite science was biology. I remember one of our first lessons the teacher gave us a diagram and asked us to place body organs where we thought they were located. My best friend put the kidneys in the brain! lol! She went on to become a staff nurse though not a kidney specialist I hope!
ReplyDeleteGreat post - love a theme - my Round 11 is all about my chickens!
Denise
ABC Team
Well, then "J" will be on biology. :-)
DeleteYes, this is the "nerd zone" (or "geek zone." I like "nerd," others prefer the term "geek.")
Anyways, welcome! And I love chickens. As I mentioned on your blog, I've always wanted to own a few Orpington hens as egglaying pets (I'm a vegetarian.)
Same here, Science wasn't my stellar subject.
ReplyDeleteThe letter E for me and science animals would have to be elephants. Seeing these magnificent animals in the wild in Kenya was a huge life moment for me.
Well, I'm glad you said something! I was going to do it on echidnas, the only egg-laying mammals other than the platypus. But if you like elephants, elephants it shall be! :-)
DeleteChris, what a fun blog! First time here, thanks to ABC. Roger grew up in my hometown, so we are sort of friends-in-law via the blogs... isn't he the best on factoids?
ReplyDeleteScience. I was not good at it. I protested when the "kill the fruit flies" experiment came around and actually staged a walkout when they euthanized frogs... but I am rather informed on the subject of mental disorders, being manic depressive and having PTSD. I think I could manage one on "bipolar" if you want - caveat is, you have to email me and remind me when it's my turn, because I also have short-term memory loss, ha ha ha. (Gallows humor flourishes in the mental health community. Without it, you're lost...)
Again, delightful post. And you would have HATED having me as a student, I'm sure! Peace, Amy
http://sharplittlepencil.com/2012/07/31/canvas-wwp-abc-poets-united/
and my email is sharplittlepencil (AT) gmail (with the dot com)
Thank you for coming!
DeleteI think you misunderstand (or I misunderstand you?), but I'm doing requests.
Any science you can think of that you like, I'll give you a letter and do a post on it. Any science at all - it's like a little challenge for me. :-)
Chemistry is always Cool. Now waiting for astronomy ;)
ReplyDeletePhenoMenon, ABCW Team
:-) I'll let you know when yours is up.
DeleteYou are very clever! Catching up with ABC.
ReplyDeleteC is for...
Rose, ABC Wednesday Team
PS.. COMMENTS makes me happy!
Thanks for stopping by!
DeleteThanks for a new word that I hope will stay in my head. Technical exercises for pianists are sometimes in Contrary and sometimes in parallel motion. Contrary motion always seemed easier to me, I guess because of the chirality factor. My favourite science was biology, but if you feel inspired, the physics of sound would be an interesting topic. I am also, along with Photowannabe, passionate about elephants. Do you know about www.elephants.com? I have followed that site ever since Tina (elephant from Vancouver) went to that sanctuary. She died a year or so later, but at least her last year of life was happy. I've also been vegetarian (changed to vegan in January) for more years than I can count. Must be a science topic in there as well. Wonderful blog and great idea to Channel the topics into a science theme.
ReplyDeleteNo, I didn't know about elephants.com, although I do like elephants. Thank you for the link.
Delete"J" is already on biology, but I bet I can do an awesome "K" on the physics of sound for you!
Looking forward to all your posts. One of the greatest things about blogging is how much you can learn. (I think it's holding off alzheimers for me!) :)
ReplyDeleteThank you for stopping by!
Delete